Aphids 101: The Small, Green World Of Family Aphidoidea
Aphids, Greenfly, Blackfly, Plant-lice etc. are pests – little blighters that suck the life out of your garden plants. Everybody knows this.
However, though aphids are often pests – the truth is that they are fascinating animals that have evolved some amazing strategies to allow them to be a numerically very successful life form.
I hope that by the time you have finished reading this you will look at aphids more openly – as you wipe them out.
Aphids are related to Adelgids (Conifer Aphids) and Phylloxerids, all of which probably evolved about 280 million years ago (MYA) in the Carboniferous. They probably bred on non-flowering plants such as Cordaitales and Cycadophyta (flowering plants i.e. Angiosperms, did not evolve until about 160 MYA).
The oldest known fossil aphid is Vosegus triassicus from the Early-Middle Triassic about 247 MYA. Aphids did not always look like they do now; the cauda and the siphunculi did not evolve until the Cretaceous about 55MYA.
There are about 4,000 species of aphids in the world, of which about 250 are serious pests. Scientists believe that the number of species remained relatively small until the angiosperms (flowering plants) became more common. Then, as the aphids adapted to these new and rapidly speciating plants, so their species numbers increased also.
Twenty-five percent of all plant species are infested with aphids; and though it is believed that the speciation of aphids has followed that of plants, not all groups of plants are equally parasitised.

The Compositae, the 3rd most specious plant family, supports the most aphid species (605 species); but the Orchidacea, the 2nd most specious plant family, only support 9 species of aphids; while the Rosaceae, which is only the 22nd most specious plant family, supports the 3rd highest number of aphid species (293 species). The plant family which supports the 2nd highest number of aphids is Coniferae (363 species), but these are non-flowering plants.
Aphids have a world-wide distribution, but there are far more species in temperate zones. We have more than 500 species in the UK, while there are about 1,350 species in North America. Most aphids are monophagous, though some feed on more than one species of plant; species like Myzus persicae, which feed on a number of different plant species, are very rare.
You may use the links below to skip directly to the relevant part of this page:
- Life Cycle
- Honeydew
- Ants and Aphids Relationship
- Polyphenism
- Migration and Dispersal
- Defense
- Predators and Parasites
- Aphids as Pests
Aphidoidae Description
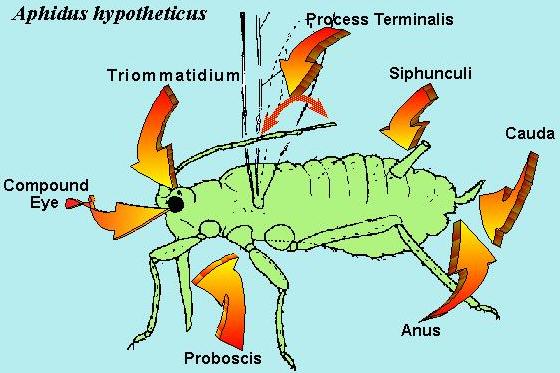
Aphids are part of the Superfamily Aphidoidae and are described as small (1-10 mm) soft bodied insects with (or more commonly, without) wings. They possess a proboscis, which originates between and behind the forelegs. Their antennae has two thick basal segments and a flagellum composed of up to 4 segments, the last of which is divided into a proximal part and a thinner distal part called a ‘process terminalis’.
They possess two compound eyes and two ocular tubercles, made up of three lenses each, which are situated behind and above the compound eyes. They have two tarsal segments; the wings when present have only one prominent longitudinal vein.
The fifth abdominal segment bears a pair of upward and backwardly pointing tubes on the dorsal surface called siphunculi (cornicles); and a cauda is usually present below and between them on the last abdominal segment.
Aphids are often parthenogenetic for part (or all) of their lives; and both viviparous and oviparous at different times of year. Parthenogenetic (asexual) reproduction generally results in viviparity (live birth) and all aphids are both parthenogenetic and viviparous during Spring and Summer.
Some aphids which are parthenogentic during the Summer produce sexual offspring (males and females) in Autumn; this is termed ‘cyclical parthenogenesis’ or a ‘holocyclical’ life cycle. The sexual males and females mate in the Autumn.
Sexual females, like asexual ones, have 2 sex chromosomes i.e. XX. Males have only one sex chromosome i.e. OX. In theory, this means males could produce sperm with either no sex chromosomes (i.e. a O) or one sex chromosome (i.e. an X). However, in reality, sperm with an O sex chromosome degenerate very rapidly and never contribute to an embryo.
This means that all offspring of a sexual mating must have XX as their sex chromosomes, because females always contribute an X chromosome; therefore all aphids resulting from sexual matings are female.

Eggs are laid during the Autumn, as the overwintering stage in many temperate forms (and as explained above) gives rise to females – whether they are the result of sexual mating or not.
In other species, a special overwintering form develops in the Autumn called a ‘hiemalis’; while in some species the adults are the overwintering stage. Ova within a viviparously reproducing female start to develop immediately after ovulation; this occurs long before birth (even human females are born with all the ova they will ever need throughout their life, though they remain undeveloped for many years).
This means that an embryo can exist inside another larger and more mature embryo.
In fact, a newly born Summer aphid can contain within herself not only the developing embryos of her daughters, but also those of her grand-daughters – which are developing within her daughters.
Parthenogenesis combined with this ‘telescoping of generations’ give aphids an exceedingly rapid turn-over of generations – meaning they can build up immense populations very quickly.
Aphids feed from the phloem of plants, which they tap into with the stylets of their proboscis. They gain access to the phloem vessels from 3 main parts of the plant: stems, leaves, and roots. Their stylets, which are contained within the proboscis when the aphid is not feeding, are very thin and could suffer damage while being pushed into the plant or bent in an unwanted direction.

Therefore aphids secrete a special liquid from the tips of their stylets, which starts to harden as soon as it leaves the stylets. This forms a hard, protective sheath around the stylets – as they are slowly pushed into the plant in search of the phloem tubes. When the stylets reach a phloem tube, the aphid injects saliva into the plant cell.
It is suspected, but not known for sure, that this saliva helps prevent the plant cell from sealing the puncture (i.e. the aphid’s mouthparts) with special proteins. These special proteins are the plants normal defence mechanism. They are deposited on the wall of the cell around a puncture, as a result of the drop in redox potential that occurs along the cell wall following puncture damage.
Aphids insert their stylets slowly and it takes quite a bit of time to tap into a phloem tube. It can be anywhere from 25 minutes to 24 hours from starting to insert the stylets, to actually getting something to eat.
Some aphids always feed by inserting their stylets through the stomata of the plant leaves they are feeding on, i.e. Ceratovacuna lanigera on Miscanthus spp. and Schizolachnus orientalis on pine needles. Other than this, aphids penetrate to the phloem either intracellularly (pushing the stylets between cells); intercellularly (pushing the stylets through cells); or by a bit of both.
Plant phloem saps are rich in sugars and poor in amino-acids or nitrogen. This results in aphids excreting large amounts of sugary liquid – called honeydew – because the amount of sap they have to drink in order to get enough nitrogen in their diet, means they have far more sugar and liquid than they need.

This honeydew can often be seen on the lower leaves of infested trees, which it falls on giving them a sticky coating. This is then fed on by other insects, such as the Common wasp Vespula vulgaris; the Brown Hairstreak Butterfly Thecla betulae; and a yeast-like fungus which makes the leaves look all black as if they were covered in soot.
Nearly all aphids contain endosymbionts in special groups of cells called bacteriocytes.
The exceptions are some members of the Cerataphidini, which have yeast cells in their haemocoel instead. In most aphids the main endosymbiont is Buchnera aphidicola, a relative of Escherichia coli which is common in our guts. Buchnera aphidicola is believed to complement the aphid’s diet by synthesising vitamins, sterols and certain amino acids.
Whatever their role, they are important for the aphid’s growth and reproductive potential; they decrease as the aphid gets older and are absent from most soldiers and males. The relationship between Buchnera and the aphids it lives with is obligatory on both sides, i.e. the bacteria can’t live outside of the aphids and the aphids can’t reproduce successfully without it.
Buchnera is transmitted maternally via the ovary, i.e. new aphids get the symbiont from their mums when they are just an egg.
Most aphids are autoecious (living on one or a few species of closely related plants). About 10% are heteroecious, spending Autumn, winter and spring on one plant species (its primary host) and Summer on a different unrelated plant (its secondary host).

For example the Rosy Apple Aphid Dysaphis plantaginae has Apple Malus sp. as its primary host and Plantain Plantago lanceolata as its secondary or summer host. Some heteroecious aphids such as Myzus persicae the Peach or Potatoe Aphid and Aphis fabae the Black Bean Aphid, have a wide range of secondary hosts; but this is relatively rare. Most heteroecious aphids have just one primary and one secondary host.
Though aphids are very successful at feeding on plants, they do not always have things all their own way. Small spines, hairs and sticky latex secretions are among the more successful methods that plants have evolved to protect themselves against these parasites.
Honeydew is the reason that ants have a symbiotic relationship with aphids.
In fact, many species of ants are so addicted to this sweet drink that they will protect the aphids from various predators and move them to new plants if the one they are on start to wilt. You may well even have heard tales about ‘ants farming aphids’.
Some ants even go as far as to build small shelters over species that feed near the base of the plant; and/or to keeping root aphids inside their own nests.
Lasius fuliginosus workers carry newly hatched fundatrices from the base of oak trees, where they overwinter as eggs, to new growing leaves at the top of the tree – as soon as they are hatched.

The common meadow ant Lasius flavus has a particularly close relationship with the root aphids it uses; it even collects their eggs in the autumn and early winter and stores them in its nests. Then in spring, the eggs are moved to suitable chambers so that plant roots are available for them as soon as they hatch. In effect these ants treat the aphids as well as they treat their own brood.
The interaction between Aphids and ants has been going for a long time and some ants are almost dependent on aphids for food, while some aphids such as Protrama spp. are obligate myrmecophiles and do not excrete honey-dew unless stimulated to do so by ants.
Species of aphids which have intimate symbiotic relationships with ants, particularly those that live in their nests, are of necessity monoecious. The aphid Paracletus cimiciformis is practically only found in the nests of Tetramorium caespitum, where it is fed and cared for by the ants – despite the fact that it rarely if ever secretes honey-dew. It is in fact a parasite and gains most of its food from the ants who offer it nectar.
The number of ants associated with a given species of aphid (and the number of aphid species associated with a given species of ant) varies from place to place; up to 17 different aphid species have been found in Lasius niger nests.

Sometimes different ant species which live in similar habitats will foster different aphid species, i.e. Lasius niger with Anoecia corni and Lasius flavus with Anoecia nemoralis. However aphid species that have evolved close relationships with ants may have broader environmental tolerances than their host’s. Hence the aphid Forda formicaria is attended by Lasius spp. in the lowlands; and by Formica spp. in the highlands.
Ants gain much nutrition from their relationship (or symbiosis) with aphids – and the honey-dew excreted by aphids such as Aulacorthrum sp. can in some cases be a complete ant food. Generally the amount of proteins and amino acids in excreted honey-dew varies between species and plant.
However because ant attendance stimulates aphids to feed at a rate 2-3 times their normal rate – and because they do not grow at 2-3 times their normal rate – it follows that the reduction of nutrients resulting from aphid digestion is greatly lessened.
Also as adult, ants do not need proteins but only sugars – honey-dew is a perfect food for them.
Ants are not always good guys though… like us with cows, if aphid numbers build up, the ants quite happily kill a few off and feed them to their larvae.
The protection that ants give the aphids they attend is not always excellent and varies depending on the type of predator or parasite. Generally speaking, the ants are better at dealing with Ladybird larvae and Anthocorid bugs, than they are at dealing with Lacewing larvae and Hover Fly larvae.

They not only remove the larvae, sometimes killing them, but also remove the eggs of hoverflies and Ladybirds. Interestingly, by herding aphids onto the tops of the plants, ants render them more vulnerable to attack from some parasites. In one experiment in 1958, R.C. Banks found that attended aphids were 10X more likely to be parasitised than unattended ones.
Aphids exhibit polyphenism; summer populations are all ‘clones’ of an original foundress or fundatrix, as a result of parthenogenetic reproduction (see above), i.e. they are all genetically the same.
But they are not all morphologically the same. In a number of species of aphids, soldiers occur – which protect the other members of the colony. These soldiers possess enlarged prehensile forelimbs and a spike on the front of their heads – which they use along with their proboscis to attack and kill potential enemies of the colony.
Soldiers are 2nd instar nymphs, do not feed or reproduce and spend their short lives solely for the protection of the colony.

Species with soldiers are normally gall-forming species such as Ceratoglyphina bambusae, which causes large galls containing 100 to 200 thousand aphids, to form on the Snowball tree Styrax suberifolia. In large colonies of Ceratoglyphina bambusae, up to 50% of the aphid population may be soldiers.
In Cerataphis fransenni, the number of soldiers per gall is proportional to the surface area of the gall and the total number of aphids present. It is interesting to note that, though soldiers will attack non-soldier aphids of the same species from a different colony (conspecific), when they encroach on their own colony’s gall they do not attack conspecific soldiers – perhaps because they do not feed or breed.
Soldiers will however attack soldiers, as well as non-soldiers of other species, if they encroach on their gall.
Aphids also occur in different colour forms or morphs. The Pea Aphid Acyrthosiphum pisum occurs in red or green morphs. Two of the aphid’s main predators are the Ladybird Coccinella septempunctata and the Parasitic Wasp Aphidus ervi.

The Ladybird prefers the red morphs and feeds preferentially on them, while the Parasitc Wasp prefers the green morph. Both tend to prefer hunting where there are lots of aphids and to move on when their preferred morph is reduced to low numbers. In this way, the morph is maintained in the population – which wouldn’t happen if one colour morph was always more attractive to both or all predators.
Most of the time, aphids do not have wings; in general they move very little, and life consists of feeding and giving birth. However, four stimuli cause an aphid to move and two of these really only work on the population level.
The first two stimuli to movement are the death of the host plant (which stimulates aphids to walk away from the host plant in search of a new one) and/or danger (see below).
The other two are environmental changes (signalling that it is time to move, either to the primary or secondary hos)t and overpopulation (which stimulates aphids to migrate away from their present area).

Both these stimuli result in the production of alatae (possessing wings) forms appearing within the colony/population. It is these winged forms that do the moving, either to a new host plant or as a migration to new areas where fresh hosts can be found.
On their own, aphids are weak fliers and in still air they move at about 1.6 to 3.2 km per hour.
Their migrations can be quite extensive and they often take advantage of favourable winds to enhance their flight efficiency. Aphids which fly upwards to get above the planetary boundary layer (PBL) early on in the night, can end up more than 1,000 metres above sea level and are carried by the low level jet streams that occur at these heights.
This can result in them travelling more than 400 km in 9 hours.
They descend again in the early dawn, as sun warming the surface of the planet causes updrafts which disrupt the PBL and make air travel much more dangerous. When coming into land from these great heights, the aphids home in on leaves reflecting long-wave light.
They are particularly attracted to the yellowish light emitted from young, actively growing crops or older senescing ones.
This is because the flow of nutrients to and from the leaves is greater for leaves at these stages of growth than for ordinary green leaves.

Several pest species are recorded as long distance migrants in North America; These include the English Grain Aphid Macrosiphum avenae; the Corn Leaf Aphid Rhopalosiphum maidis; the Oat Bird-Cherry Aphid Rhopalosiphum padi; and the Greenbug Schizaphis graminum.
The last and most common stimulus is attack by a predator or parasite. An aphid which is which is under attack can defend itself by a variety of means, depending on the size and nature of the attack.
First off, an aphid will kick out at an attacker and attempt to drive it off; if this does not work, it will pull up its stylets and proboscis and walk away.
Some species can also exude a special waxy solution from their siphunculi over an attacker. While doing this, it generally releases an alarm pheromone alerting nearby aphids that danger is close at hand.
An aphid’s final solution is to jump or fall of the plant. This of course necessitates the aphid climbing back up onto the host plant again, which if it is a tree can be quite a climb.
Aphids are relatively common and in the right circumstances they occur in large numbers. For this reason there are quite a number of animals which use aphids as a food source. Many of these specialise on aphids; some of these are listed here; others are general predators which take advantage of aphid colonies when they can, these are listed below.
Obligate Aphid predators and parasites.
- Hoverfly larvae (Syrphidae); a number of species particularly in the Syrphinae, i.e. Syrphus ribesii and Episyphus balteatus.
- Aphid Midge larvae (Cecidomyiidae); i.e. Aphidoletes aphidomyza;
- Flower Bugs (Anthocoridae); i.e. Anthocoris confusus. Some Anthocoris species live and feed on only one genus of aphids, i.e. Anthocoris gallarum-ulmi feeds only on Schizoneura sp. on elms.
- Lacewings (Neuroptera); i.e. Chrysoperla carnea. Lacewings normally only eat aphids as larvae, but some adult Hemerobiidae will take aphids.
- Chamaemyiidae; Leucopsis spp. larvae.
- Chloropidae; Thaumatomyia spp. larvae, specialise on root aphids.
- Phoridae; Phora spp. larvae, specialise on root aphids.
- Solitary Wasps (Specidae); Passaloecus spp. the adult females stock their cells of their larvae with aphids.
- Parasitic Wasps (Parasitica); there are many of these and some are specialists on aphids and some even on only one genus. Some examples are the Braconids (Braconidae) Monoctonus pseudoplatini and Trioxys cirsii; the Aphelinid (Aphelinidae) Aphelinus flavus, all on the Sycamore Aphid Drepanosiphon plantanoides. Other examples are the Braconids Xenostigmus bifasciatus on Cinara spp., Ephedrus californicus on the Pea Aphid Acyrthosiphum pisum, Aphidius sonchi on the aphid Hyperomyzus lactucae and Aphidius nigripes on Macrosiphum euphorbiae.
Facultative Predators and Parasites
There are many predatory insects which will eat aphids, if given the chance. These include:
- Earwigs (Dermaptera),
- Bush Crickets and Katydids (Orthoptera) especially in the early instars,
- The Hemipterans: Damsel Bugs (Nabidae) and Mirid Bugs (Miridae),
- The Coleopterans: Ground Beetles (Carabidae) and Rove Beetles (Staphylinidae),
- The Dipterans: Stilt Legged Flies (Micropezidae) and various members of the Dolichopodidae, Empidae and
- Hymenopterans: Ants (Formicidae) and Wasps (Vespidae).
Apart from these, aphids are also consumed by various Spiders (Araneae) and Harvestmen (Opilones).
Aphids are pests… at least about 10% of them are (of which only about 250 are considered serious pests).
They are pests firstly because their feeding reduces the vitality of the crops they feed on; and secondly because in some cases they transmit viral diseases.
Perhaps the most important aphid pest is Myzus persicae the Peach-Potato Aphid. M. persicae is a green or slightly reddish aphid, which has Peach as its primary host and a wide range of secondary hosts including many brassicas.

M. persicae is cosmopolitan in temperate climates, occurring in USA and a fair portion of Europe including the UK. Though it seldomly occurs in numbers large enough to cause direct damage from feeding pressure, it is capable of transmitting/spreading over 100 viruses including: the Potato Leaf Roll and Y viruses of potatoes; Mosaic, Yellow net and Yellows viruses of Sugar-beet Cauliflower Mosaic Plum Pox, Cucumber Mosaic, Lettuce Mosaic, and Turnip Mosaic.
See below for some of the most serious aphid pests:
The Black Bean Aphid (Aphis fabae)
The Black Bean Aphid Aphis fabae is another major pest, which can if unchecked cause losses of up to 46% on Broad Bean crops. Its primary host is Spindle Tree Euonymous europaeus, where eggs are laid in the axils of buds and in crevices in twigs – but population numbers only build to infestation level on the summer crop hosts.

It does not reach epidemic levels every year, and it is possible to forecast the likelihood of infestation (and the need to spray chemicals) by monitoring the numbers of aphids flying away from the primary host in spring. For this to work properly, a large number of suction traps need to be being operated around the country.
Forecasting likely pest levels is an important science because it can greatly reduce the amount of pesticides that need to be spread around the country.
Unfortunately, for uncertain reasons, many of the suction traps in the UK are not being run as much as they were – and forecasting is not being properly used.
The Pea Aphid (Acyrthosiphum pisum)
The Pea Aphid Acyrthosiphum pisum, is a large green aphid with long antennae and legs. The Pea Aphid is found on many leguminous plants and transmits Lucerne mosaic virus, pea leaf roll virus, pea Enation mosaic virus and pea mosaic virus in the UK; and pea Enation mosaic virus in the USA. The Pea Aphid has only one host.
In colder areas, eggs are laid by sexually mated females in overwintering forage crops and hatch early in the spring to found new colonies; which, as they grow in size, produce winged individuals that disperse to other crops. In warmer climes, overwintering is by asexual females and no sexuals occur.
Cabbage Aphid (Brevicoryne brassicae)
The Cabbage Aphid Brevicoryne brassicae is a serious pest of the major cabbage crops; Cabbages, Cauliflowers and Brussel Sprouts. Like the Pea Aphid, the Cabbage Aphid is a one host species – spending all its life on Brassicas.

The main overwintering form is eggs from sexual matings, but in warmer years asexual females may successfully overwinter as well. The eggs hatch in February or March, so the species gets off to an early start. The main cause of its pest status is the transmission of the viruses Cauliflower Mosaic and Turnip Mosaic.
More Aphid Pests:
- Grain Aphid Sitobion avenae
- Carrot Aphid Cavariella aegopodii
- Groundnut Aphid Aphis craccivora; a polyphagus species that prefers legumes.
- Cotton Aphid Aphis gossypii; polyphagus in warmer regions.
- Black Citrus Aphid Toxoptera aurantii; polyphagus in warmer regions.
- Willow Aphid Cavariella spp. on willows, Holarctic
- Willow Leaf Aphids Chaitophorus spp. on Willows and Poplars in europe
- Black Pine Aphids Cinara spp. on a variety of conifers widespread in America, Africa, Asia and europe.
- Sycamore Aphid Drepanosiphum platanoides on Sycamore only in Europe and North America.
- Spruce Aphids Elatobium spp. on Picea in North America and Europe.
The control of aphid pests currently still involves large amounts of pesticides in some countries.
But other more ecologically friendly methods have been used in other places for some time. These generally involve biological control (mostly with a range of Hymenopteran parasites); genetic manipulation for aphid resistance; and forecasting to allow for pre-emptive spraying in smaller amounts.
All these are being studied in more depth and will become more functional as time moves on, so that more detailed information is easily made available to the forecasters and farmers can be more readily educated on the alternatives to chemical sprays.
Special (Aphid) Terminology
- Alatae = Winged female, Summer migrant.
- Apterae = Wingless female.
- Fundatrix(rices) = A clone/colony foundress.
- Monoecious = having only one host plant, i.e. feeding on one plant species all the time.
- Heteroecious = having a primary and a secondary host plant.
- Holocyclic = Cyclical parthenogenesis = reproducing by means of sexuals in autumn and by parthenogenesis for the rest of the year.
- Anholocyclic = reproducing only by parthenogenesis.
- Sexupara = an aphid that gives birth to male and female sexuals.
- Gynopara = a winged sexupara who gives birth to egglaying females only.
- Ovipara = the female sexual capable of mating and laying eggs.
- Virginopara = winged or unwinged aphids which give birth parthenogenetically to live young.
- Parthenogenesis = reproductive production of females by unmated females from unfertilised eggs.
- Viviparous = giving birth to live young.
- Oviviparous = giving birth by laying eggs.
Aphidoidea Taxonomy
Aphids are divided up into 10 families, all of which have been extant for 50 to 70 million years. Two families, the Aphididae and the Drepanosiphidae, make up 70% of the species.
Superfamily Aphidoidea
- Pemphigidae
- Anoeciidae
- Hormaphididae
- Mindaridae
- Thelaxidae
- Drepanosiphidae
- Phloeomyzidae
- Greenideidae
- Aphididae
- Lachnidae
What Next?
Well, I hope this has been an interesting glimpse into the incredible world of superfamily Aphidoidea!
Perhaps now you’d be curious to learn more about the wonders of cicadas.
Bibliography
- Aphids, their Biology, Natural enemies and Control, (1987) by P. Harrewjin and A. Minks amsterdam, Elsevier.
- The Aphididae of North-West India (With Special Reference to Aphids of Jammu and Kashmir State), by K. D. Sharma and S. M. Das (Eds)
- Aphids on the World’s Trees. An Identification and Information Guide, by R. L. Blackman and V. F. Eastop
- Atlas of Aphids Trapped in Agricultural Crops, by L. L. Gualtieri and D. G. R. McLeod
- The Insects and Arachnids of Canada. Part 22. The Genera of the Aphids of Canada: Homoptera: Aphiodea and Phylloxeroidea, by RG Foottit and WR Richards
- Population Structure, Genetics and Taxonomy of Aphids and Thysanoptera, by J. Holman, J. Pelikan A. F. G. Dixon and L. Weismann (Eds)
- RES Handbook Volume 2 Part 6 Hemiptera – Aphids Out of Print
- Aphids of Japan in colors, by M.Moritsu. (1982), Tokyo Zenkoku Noson
- Aphids of Korea, by W. H. Paik. (1965), Seoul, Seoul National University
- A Catalogue of Aphids (Homoptera: Aphidoidea) of Sub-Saharan Africa, by IM Millar
- Aphidoidea (Insecta) of Latvia. Rupais A. A. 1989.Riga: Zinante
- Types of adaptations of Aphidoidea (Insecta) to feeding on different plants. Popova A. A. 1967.Leningrad: Nauka,
- Aphidoidea (Insecta) of West Siberia. Pt. 1. Adelgidae-Chaitophoridae. Ivanovskaya O. I. 1977. Novosibirsk: Nauka,
- Biology of Aphids, by j.S. Kennedy and H.L.G.Stroyan, (1959) Annual Review of Entomology, 4 155-174.
- Mutualism between ants and Honeydew-Producing Homoptera, by M. J. Way, (1963) Annual Review of Entomology, 8 307-344.
- Aphid Feeding and Nutrition, by J. L. Auclair (1963) Annual Review of Entomology, 8 439-490.
- The Ecology of Myzus persicae, by H. F. van Emdem, V. F. Eastop, R.D. Hughes and M. J. Way. (1969) Annual Review of Entomology, 14, 197-270.
- Aphid Ecology: life cycles, polymorphism and population regulation, by A. F. G. Dixon, (1977) Annual Review of Ecological Systems, 8, 329-353.
- Structure of aphid Populations, by A. F. G. Dixon, (1985) Annual Review of Entomology, 30 155-174.
- The Chemical Ecology of Aphids by J. A. Pickett, L. J. Wadham, C. M. Woodcock and J. Hardie, (1992), Annual Review of Entomology, 37 67-90.




